 Bond angles: The angle between adjacent bonds of an atom.Hybridization: Orbitals are combined in order to spread out electrons.Molecular Geometry: Describes the arrangement of atoms around the central atom with acknowledgment to only bonding electrons.Electron Geometry: Describes the arrangement of bonds and lone pairs around a central atom.If you enjoy this tutorial, feel free to check out our other tutorials on bonding listed below. You will learn about the more common molecular geometries: tetrahedral, linear, bent, trigonal pyramidal, and trigonal planar – along with their bond angles. Water has four electron groups, but only two atoms attached to the central atom so it is bent.In this tutorial, you will learn how to identify the molecular geometry and bond angles of a molecule. First draw the Lewis electron dot diagram for water and determine its molecular shape. The first two steps remain the same as the tail-to-head method: 1. Let’s examine this method again for a molecule of water. An alternative method to determine the vector sum of dipole arrows is known as the vector component method. Now superimpose the net molecular dipole arrow onto the molecule. Draw a new line connecting the tail of the first vector. Draw in dipole arrows for all polar covalent bonds, starting the arrow at the more electropositive atom, and ending at the more electronegative atom. Water has four electron groups, but only two atoms attached to the central atom so it is bent. Let’s examine this method for a molecule of water. One method to determine the vector sum of dipole arrows is known as the tail-to-head method. Therefore the molecular polarity is the vector sum of the individual bond dipoles. Each bond’s dipole moment can be treated as a vector quantity, having a magnitude and direction. 
The overall polarity of molecules with more than one bond is determined from both the polarity of the individual bonds and the shape of the molecule. Table 9.4 Summary of Molecular Shapes Number of Electron Groups on Central Atom When the two electron groups are 180° apart, the atoms attached to those electron groups are also 180° apart, so the overall molecular shape is linear. A molecule whose central atom contains only two electron groups orients those two groups as far apart from each other as possible-180° apart. Remember that a multiple bond counts as only one electron group.Īny molecule with only two atoms is linear. When applying VSEPR to simple molecules, the first thing to do is to count the number of electron groups around the central atom. There are two types of electron groups: any type of bond-single, double, or triple-and lone electron pairs. 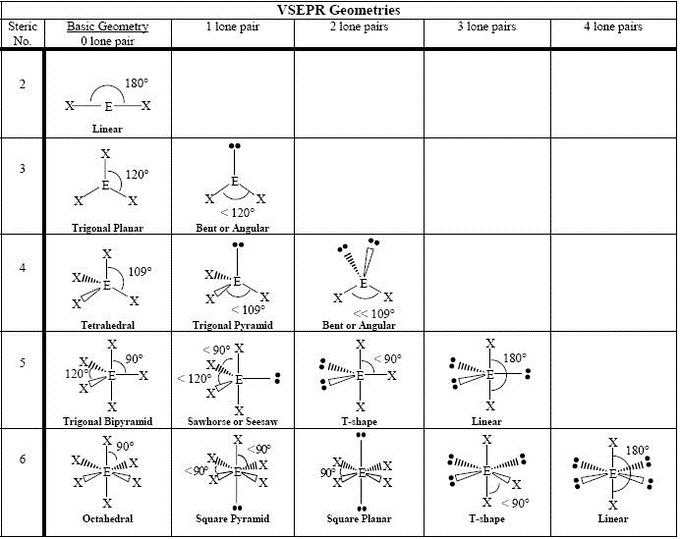
VSEPR makes a distinction between electron group geometry, which expresses how electron groups (bonding and nonbonding electron pairs) are arranged, and molecular geometry, which expresses how the atoms in a molecule are arranged. It says that electron pairs, being composed of negatively charged particles, repel each other to get as far away from each other as possible. The basic idea in molecular shapes is called valence shell electron pair repulsion (VSEPR). Small molecules-molecules with a single central atom-have shapes that can be easily predicted. There is an abundance of experimental evidence to that effect-from their physical properties to their chemical reactivity. Determine the polarity of molecules using net molecular dipoles.Determine the shape of simple molecules. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
